Perspectives: Orgenesis, Inc.
Nano-captivating prospect is industrializing cell and gene therapies
Perspectives is a special feature in Confluence. We occasionally highlight emerging growth companies that capture our attention and whose progress will continue to be monitored. Note that these briefs reflect our initial research on the company and are not complete analyses or recommendations.
Waves of IPOs for early-stage biotech and biopharma companies flooded the public market from 2001 to 2022. Jay R. Ritter of the University of Florida counts 632 biotech/biopharma IPOs—after excluding those with an offer price below $5.00 per share. This year, so far, only a handful managed to complete new issuances.
The bear market in small biotech stocks, now almost two years old, has washed away market value in hundreds of companies. Of the 544 biotechnology companies tracked by Third Stream Research, 248 (~46%) are down at least 50% over the past year (as of 5/1/23), two-thirds of which are currently nanocaps (<$50M market cap). Recent issues of Confluence have scrutinized the damage, most notably Biotech Retrenchment in January.
Selloffs of this kind tend to go to extremes. We believe that the massive drawdown in microcap and nanocap biotechs since mid-2021 was too indiscriminate in recalibrating valuations. Stature of drug pipelines and cash burn rates are rightly big factors: so many companies are either already in the intensive care unit or unrealistically biding their time before they end up there. But surely, we thought, there must be compelling prospects worth a serious look at such nominal valuations.
Two prospects caught our attention as we began to sift through the wreckage. The first, Orgenesis, Inc. is seeking to change the business model around cell and gene therapies through its point of care platform, which it says can lower the cost, accelerate development and expand access for patients. Importantly, it is a scalable, high margin, recurring revenue business model.
Orgenesis, Inc. (ORGS)
Price (5/5/23): $1.07
52-Week High: $3.28
52-Week Low: $0.98
Avg. Daily Volume: 79,400 shares
Market Capitalization: $29.5M
Enterprise Value: $47.2M
Shares Outstanding: 27.9M
Free Float: 25.3 million
Insider Ownership: 12.0%
Institutional Ownership: 11.1%
Short Interest: 327,000 shares
Revenue (LTM): $36.0M
EBITDA (LTM): ($5.5M)
Net Income (LTM): ($14.9M)
Long-term Debt: $13.3M
Cash: (12/31/22): $5.3M (raised $8.9M since year-end)
Addressing shortage of production capacity in the cell and gene therapy space
The gene therapy industry has come a long way in the past decade, with thousands of products currently under clinical development or approved for use. According to a report by Grand View Research, the global cell and gene therapy market size was valued at $7.8 billion in 2020 and is expected to grow at a CAGR of 33.3% from 2021 to 2028. By 2028, the market size is projected to reach $21.0 billion.
Despite the immense potential for growth, two significant challenges continue to hinder the industry's progress: cost and capacity.
Optimizing manufacturing methods and increasing industry-wide capacity could alleviate the high costs of cell and gene therapies. According to the Institute for Clinical and Economic Review, gene therapies can cost between $1 million and $2 million per dose, with the average cost per cell therapy treatment at $1 million. The complexity of producing, handling, and controlling the cells or viral vectors required to make these therapies is largely responsible for their high cost. Developers consider factors such as R&D investment, market demand, competition, and manufacturing cost when setting prices.
Equally important is that biotech organizations struggle to produce enough of the stuff to meet demand. Even when life-saving therapies are approved, the limited number of patients who can access them highlights the issue of capacity. This is especially true for autologous products, which require the patient's cells to be collected and transported to processing facilities, often in distant locations, before being sent back to the patient.
Additionally, there are still regulatory hurdles that need to be overcome. The approval process for cell and gene therapies is complex and time-consuming, which can make it difficult for companies to bring their products to market. There are also concerns around safety and efficacy, which need to be addressed through rigorous clinical testing.
Finally, there are infrastructure challenges associated with the manufacturing and distribution of cell and gene therapies. Many of these therapies require specialized facilities and expertise to produce, which can limit their availability and increase their cost. These challenges need to be addressed in order to ensure that cell and gene therapies can reach their full potential and benefit patients worldwide.
The crux of these issues is how to industrialize gene therapies. While progress has been made, there's still a long way to go to make these therapies more accessible and affordable for patients.
Reprogramming cells: a new approach to biotech manufacturing
The biotech industry has long attempted to fit emerging modalities into existing paradigms of good manufacturing practices, sterile environments, and centralized facilities. However, the development of cell therapies, largely by clinicians and university researchers, has exposed the limitations of this approach. Traditional biotech does not create a factory or manufacturing facility per patient, making the centralization of these therapies expensive and inefficient.
Orgenesis built a centralized contract development and manufacturing organization (CDMO), Masthercell, growing it from $3 million to over $33 million in revenues from 2014 to 2019. ORGS sold it for $315 million in February 2020 with net proceeds of $127 million, which served to fund its transition into its current business model—a three component POCare Service Platform for novel cell and gene therapies to reach patients at the point of care. The company generated more than $30 million in annual revenue within two years (2020-2021). In 2022, ORGS reported total revenue of $36.0 million.
Orgenesis has taken a radical new approach to manufacturing cell therapies. Recognizing that cells are already expertly adapted by nature to function, the company has shifted from the traditional manufacturing approach to one of cell reprogramming. Rather than breaking cells into pieces and reassembling them, Orgenesis makes small adjustments to cells’ natural code, programming them to perform specific functions.
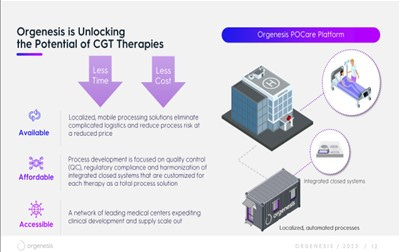
This approach requires a very different manufacturing process, one that is more akin to computing. Orgenesis has built a concept that centers around programming cells, treating biology and equipment as one unit. By unitizing the therapy, ORGS can create a robust system targeted and completely built for a specific therapy. This system can be put into an Orgenesis Mobile Processing Unit and Lab (OMPUL), and duplicated at the same or any other required location.
To reduce costs and the logistics and risks associated with transferring living biological materials to and from processing facilities, Orgenesis has set up OMPULs close to the point of care location. As a central part in the POCare Platform, these units allow the closed production of cellular therapies in a streamlined manner. They are mobile production units about the size of a shipping container.
Consequently, these OMPULs can be located and relocated easily, and a customer pays a per-batch fee. Orgenesis CEO Vered Caplan describes a batch as “taking a patient’s cells, doing the engineering, packaging, and sending the product back to that same patient.”
With these OMPULs, a healthcare provider or research group can quickly increase capacity, without the risk of building a new facility. Orgenesis allows flexibility in supply and reduce the complexity and the logistics, which reduces the cost. ORGS has created a highly innovative decentralized model that is far more scalable and cost-effective than the traditional CDMO (contract development and manufacturing organization) model.
The POCare Services that Orgenesis and its affiliated entities perform include:
Process development of therapies, process adaptation, and optimization inside the OMPULs, or ‘OMPULization’;
Adaptation of automation and closed systems to serviced therapies;
Incorporation of the serviced therapies compliant with GMP in the OMPULs that ORG designed and built;
Tech transfers and training of local teams for the serviced therapies at the POCare Centers;
Processing and supply of the therapies and required supplies under GMP conditions within ORG’s POCare Network, including required quality control testing; and
Contract Research Organization (CRO) services for clinical trials.
The POCare Services are performed in decentralized hubs that provide harmonized and standardized services to customers, or POCare Centers. Orgenesis is working to expand the number and scope of its POCare Centers. Management believes that this provides an efficient and scalable pathway for CGT therapies to reach patients rapidly at lowered costs.
POCare Services are designed to allow rapid capacity expansion while integrating new technologies to bring together patients, doctors and industry partners with a goal of achieving standardized, regulated clinical development and production of therapies.
Until 2022, Orgenesis made significant investments in R&D services including in the development of several types of OMPULs, the development of automated processing units and processes, owned and licensed advanced therapies to enable commercial production, and additional work that addresses POCare needs. While the company continues to invest in these activities, the majority of development work on its OMPULs has been completed thus allowing it to deploy OMPULs in various worldwide locations.
As a result of the reduction in development and R&D services expenses, subcontracting, professional and consulting services, lab expenses and other R&D expenses declined by 26% in 2022 from 2021.
With OMPULs already deployed across North America, Europe, Asia, and the Middle East, Orgenesis is demonstrating that its innovative approach is potentially one of the ways that lifesaving treatments will be delivered to patients worldwide, particularly in regions where high-quality healthcare is less available.
Morgenesis: Orgenesis’ subsidiary, manufacturing cell therapies for other companies and startups
In August 2022, Orgenesis formed Morgenesis LLC, a subsidiary to hold substantially all the assets of the company’s POCare Services. Morgenesis streamlines all existing POCare Service business units into one unified entity, bringing together a full-service range of solutions for therapeutic developers for point of care treatments.
Morgenesis—based at the Maryland Center for Cell Therapy Manufacturing at the Johns Hopkins East Baltimore medical campus—provides solutions from initial process development, regulatory strategy and implementation, ‘OMPULization’ which includes cGMP process development, closing/automating the process, and with the end goal of optimizing full cGMP processing and supply of therapeutic product to patients at the point of care.
Orgenesis currently owns 76.9% of Morgenesis. In November 2022, Metalmark Capital Partners, a leading private equity firm with extensive expertise in the healthcare sector, invested up to $50 million in Morgenesis LLC. The investment was made at a pre-money valuation of $125 million.
This agreement consisted of an upfront investment of $30 million and two future investments of $10 million each. The initial investment of $30 million consisted of $20 million in cash and conversion of the convertible loan Metalmark previously provided to Morgenesis in August 2022. Metalmark was founded to independently manage the Morgan Stanley Capital Partner funds.
Orgenesis transferred the following subsidiaries to Morgenesis, and the proceeds of the investment will generally be used to fund the activities of Morgenesis and its consolidated subsidiaries.
Orgenesis Maryland LLC, which is the center of POCare Services activity in North America and is currently focused on setting up and providing POCare Services and cell-processing services to the POCare Network.
Tissue Genesis International LLC, which was formed in Texas in 2022, is currently focused on development of technologies and therapies.
Orgenesis Services SRL, which was incorporated in 2022 and is currently focused on expanding the POCare Network in Belgium.
Orgenesis Germany GmbH, which is currently focused on providing CRO services to the POCare Network.
Orgenesis Korea Co. Ltd., which is a provider of cell-processing and pre-clinical services in Korea. The company owns 94.12% of the Korean Subsidiary.
Orgenesis Biotech Israel Ltd., which is a provider of process development and cell-processing services in Israel.
In December 2022, Morgenesis gained control over Theracell Laboratories, a Greek company currently focused on expanding the POCare Network.
Following the Metalmark investment in November 2022, Orgenesis separated its operations into two operating segments.
Morgenesis: includes mainly POCare Services and the results of the subsidiaries transferred to Morgenesis.
Therapies: includes Orgenesis’ therapeutic development operations.
Prior to that, Orgenesis conducted all of its operations as one single segment.
Network of partners who share expertise and technology
Orgenesis also collaborates with technology manufacturers such as Lonza—a Swiss multinational manufacturing company for the pharmaceutical, biotechnology and nutrition sectors—to develop technologies for processing cell therapies, including systems for distance therapies and quality control testing. These technologies are fitted into Orgenesis' mobile processing units, creating a network of partners who share expertise and technology.
Orgenesis' therapeutic partnerships take two approaches. The company:
Partners with developers of existing therapies, providing the industrial know-how and process development required to fit the therapy into Orgenesis’ mobile processing units. The therapy is then supplied wherever needed, while the partners continue their clinical work.
Licenses a therapy developed by a third party and provides all the industrial stages required to make it available to other hospitals and companies.
Many research institutions and hospitals are struggling to get products or industrial services from service providers or CDMOs and lack the financial capability to build up their own processing facility. Orgenesis provides a framework that enables these institutes to work together and share expertise and technology. By doing so, the development process is expedited, and the entire industry benefits from the collective knowledge and expertise.
Orgenesis’ point of care platform includes hubs located in different countries that ensure standardization of processes and quality assurance. This is critical when providing the same therapy in different locations worldwide. CEO Vered Caplan describes Orgenesis as an open source biotech company, sharing information with all of its partners and creating a network of united entities in the cell and gene therapy industry.
In March 2023, Orgenesis signed a non-binding memorandum of understanding with University of California, Davis. The goal is to progress towards a wider definitive agreement pursuant to which the university and Orgenesis POC CA aim to roll out Morgenesis’ proprietary OMPULs throughout universities within the State of California in a staged approach.
UC Davis had originally signed a collaboration agreement to join Orgenesis’ POC Network in January 2020. UC Davis Health has been utilizing Orgenesis’ POCare platform to develop, commercialize and supply cell and gene products and therapies. The agreement involved scaling up and integrating UC Davis’ lentiviral vector process as part of the Orgenesis POCare Service Platform for localized development and processing of cell and gene therapies for treating patients.
Orgenesis anticipates that this partnership with UC Davis and the universities across California will provide decentralized development and manufacturing of CGTs across a major US healthcare state.
Orgenesis’ POCare therapies: deep therapeutics pipeline in various stages of development
Orgenesis’ POCare Network is an alternative to the traditional pathway of drug development. Orgenesis works closely with many such institutes and is in close contact with researchers in the field. The partnerships with leading hospitals and research institutes gives ORGs insights into developments in the field, market potential, regulatory landscape and optimal clinical pathway to market.
The ability to produce these products at low cost allows for an expedited development process; the partnership with hospitals around the globe enables joint grants and lower cost of clinical development. The POCare Therapies division reviews many therapies available for out licensing and selects the ones which they believe have the highest market potential, can benefit the most from a point of care approach and have the highest chance of clinical success.
The goal of this in-licensing is to quickly adapt such therapies to a point-of-care approach through regional partnerships, and to out-license the products for market approval in preferred geographical regions. This approach lowers overall development cost, through minimizing pre-clinical development costs incurred by Orgenesis, and through receiving of the additional funding from grants and/or payments by regional partners.
Most new therapies arise from academic institutes or small spinouts from such institutes. Though such research efforts may manage to progress into a clinical stage, utilizing lab based or hospital-based production solutions, they lack the resources to continue the development of such drugs to market approval.
Orgenesis’ cell and gene therapies pipeline includes investigational therapies and next-generation technologies with the potential to transform the way cancer and other unmet clinical needs are treated. Its pipeline is predominantly comprised of personalized autologous cell therapies, implying that patients receive cells that originate from their own body, virtually eliminating the risk of an immune response and rejection.
Orgenesis’ pipeline focuses on advanced therapy medicinal products originated from internal proprietary, joint ventures and in-licensing agreements with biotech companies and leading research institutes. The company’s main therapeutic fields encompass cell-based immuno-oncology, cell-based drug delivery platforms, regenerative medicine, anti-viral and autoimmune disease.
Notes:
1. KYSLECEL® (Autologous Pancreatic Islets) - The patient’s own pancreatic islets, comprised of the cells that secrete insulin to regulate blood sugar, form KYSLECEL, a minimally manipulated autologous cell-based product, produced according to current good tissue practices available in the U.S. and regulated by the FDA. The target population of KYSLECEL is chronic or acute recurrent pancreatitis patients after total pancreatectomy, who are in need of insulin secretory capacity preservation. To gain insight into KYSLECEL patient outcomes, an observational study is expected to be initiated in the U.S. In addition, to promote process development and marketing of KYSLECEL in the European Union, substantial efforts are being invested. In this regard, designated teams are being trained by Orgenesis, to manage the introduction of KYSLECEL into new markets by supporting tech transfer, as well as working on the automation of the manufacturing process. ORGS is also considering new potential indications, as well as promoting the development of new additional biological product.
2. KT-DM-103 and KT-CP-203 (3D-Printed Pancreatic Islets) - Orgenesis, through the acquisition of Koligo, has exclusively licensed patents and technology from the University of Louisville Research Foundation, related to the revascularization and 3D printing of cells and tissues intended for transplantation (“3D-V” technology platform). Utilizing this technology, potential autologous and allogeneic pancreatic islet transplants may be implemented to treat type 1 diabetes (KT-DM-103), and chronic pancreatitis (KT-CP-203). In addition to pancreatic islet transplantation, the 3D-V technology platform may also support improved transplantation of other cell and tissue types.
In 2022, Orgenesis’ subsidiary Koligo Therapeutics supplied Kyslecel to five medical institutes supplying total pancreatectomy - islet auto transplant (TP-IAT) cases. Production from this one site enabled Koligo to achieve positive cash flow from operations in Q4 2022 for the business unit.
In October 2020, Orgenesis had acquired all of the outstanding stock of Koligo from its shareholders for approximately $14.5 million in shares of ORGS common stock valued at $7.00 per share and an assumption of $1.9 million in liabilities.
In February this year, Koligo received confirmation of its infrastructure design and production protocols following a successful inspection by the FDA of its registered tissue production establishment. This positive outcome has led to plans for site expansion in both the U.S. and internationally, with an eye towards satisfying the increasing demand for Kyslecel.
Leveraging the expertise of Orgenesis, Koligo is now focused on developing regional production capacity in the EU and utilizing grants to drive clinical development towards marketing approval. The move towards international expansion is expected to bring new growth opportunities to Koligo and further enhance its market position.
Orgenesis CEO Vered Caplan points out that Orgenesis does not prioritize any of these indications themselves. Rather, ORGS relies on input from the network of research institutions, hospitals, and biotechs with whom it partners.
Orgenesis licenses in therapies that are in demand from these partners, with a focus on those that can benefit from its POC platform and other network resources to help make them more available and affordable. By facilitating co-development among multiple partners, Orgenesis optimizes the chances of success for each therapy in its pipeline. Co-development partnerships help pay for development, revenue-sharing and royalty agreements with partners as the respective therapies advance through commercialization.
Financial outlook: a focus on capacity expansion
Orgenesis has three main revenue streams: POCare development services, cell process development services, including hospital supplies, and POCare cell processing.
For Q4 2022, ORG’s revenue increased by 97.7% to $13.6 million, compared to $6.9 million for the three months ended December 31, 2021. Revenue for full-year 2022 rose to $36 million, compared to $35.5 million for 2021.
Growth in Orgenesis’ latest quarterly revenue reflects an increase in cell process development service and hospital services as a result of having signed new process development services agreements with third-party customers, partially offset by a decline in POCare development services now that Orgenesis completed the majority of performance obligations under the initial development contracts in 2021.
Cost of revenues, development services and R&D in the fourth quarter and last 12 months improved significantly, declining 43% and 26%, respectively, year over year.
In previous years, Orgenesis made significant investments in R&D services, including in the development of several types of OMPULs, the development of automated processing units and processes, owned and licensed advanced therapies to enable commercial production and additional work that addresses POCare needs.
As we stated earlier, while Orgenesis continues to invest in these activities, the majority of development work on the company’s OMPULs has been completed, thus allowing it to deploy OMPULs in various worldwide locations.
Orgenesis’ operating loss Q4 2022 was $627,000, a decrease of 91%, compared to $6.9 million for the same period last year. Net loss for the three months ended December 31, 2022, was $1.3 million, a decrease of 75%, compared to $5 million for the same period last year.
For the full year 2022, operating loss was $8.6 million, a decrease of 49%, compared to $16.8 million for the same period last year. Net loss for 2022 was $12.2 million, a decrease of 33%, compared to $18.1 million for the same period last year.
Orgenesis has set its sights on achieving a minimum of 50% gross profit in revenue, alongside an EBITDA of 20% to 25% for its services, according to CEO Vered Caplan. However, out-licensing of therapy operates under a different model altogether, and ORGS is patiently waiting for the upside to manifest.
Management’s goal for 2023 is to pursue growth opportunities and expand regionally, with an emphasis on increasing capacity in the US. This expansion plan is expected to drive significant growth for Orgenesis in the coming year.
Orgenesis ended 2022 with cash and cash equivalents of ~$5.3 million and restricted cash of $1.1 million. The company subsequently raised gross proceeds of $3.9 million through a registered direct offering and a $5 million three-year convertible note, which management believes provides it additional flexibility to support its near- and long-term capital needs at the parent company level. The investment from Metalmark is expected to enable the deployment of Orgenesis’ OMPULs in the U.S. and worldwide.
A nanocap with unrecognized achievements, big growth prospects
Orgenesis is pursuing an ambitious strategy for a small, relatively unknown company in the biotech industry. Yet it’s not a newcomer. The company’s management team is experienced and has a long track record, which includes its $315 million sale of its Masthercell business in February 2020 and successful acquisition of Koligo last year.
Importantly, ORGS owns 77% of Morgenesis, which had a pre-money valuation of $125 million based on the investment by its partner Metalmark. This alone equates to an approximately $96 million value for ORGS—triple the size of its market capitalization and double its enterprise value.
Notably, production from a single site for ORGS’ Koligo—which supplied Kyslecel to five medical institutes—enabled this business unit to achieve positive cash flow from operations in Q4 2022. While Orgenesis’ full therapeutics pipeline is difficult to assess since most of the therapies are pre-clinical or early stage, it appears to compare favorably to the company’s biotech peers.
As the chart below shows, ORGS shares have trended downward since late 2021, which correlates with the bear market in small biotechs. Despite its noted achievements and improving fundamentals (e.g. revenue growth, shrinking operating losses), Orgenesis’ stock price and market cap have declined significantly.
Orgenesis runs a tight operation in serving its expansive global activities. The company’s leadership has done a good job containing dilutive financings and believes that much of ORGS’ upfront investments are behind. Even better, it expects that future revenue growth will offset the company’s cash burn.
With a current enterprise value under $50 million, we believe ORGS is an intriguing prospect with potentially big growth prospects. Indeed, this situation merits further investigation by us.
See you next week, and thank you for your support.
Josh
Connect with me on Twitter and LinkedIn.
Disclaimer
The content provided in this newsletter is intended to be used for informational purposes only. It is important to do your own analysis before making any investment based on your own personal circumstances. You should take independent financial advice from a professional in connection with, or independently research and verify, any information that you find on our website and wish to rely upon, whether for the purpose of making an investment decision or otherwise. I was not compensated by Orgenesis or any affiliates for writing and publishing this brief and do not own shares of ORGS or any other companies mentioned.